Abstract:
This project was meant to answer whether Light Emitting Diodes (LEDs) obey Ohm's Law. Given that Ohm's law would mean that voltage and current would be proportional, in oder to produce a constant resistance, we hooked up yellow and red LEDs in series with a power supply, varying the voltage and recording its effect on the current through the LED. We then graphed the data with current as the dependent variable and voltage as the independent variable. We repeated this for several different times. If the bulbs followed Ohm's law, the relationship between current and voltage would be linear, with the slope being the resistance; however, the data we produced showed the current increasing at an increasing rate; indicitive of a more exponential rate of change. We also encountered several persistent problems in data collection, including sporadic increases in current and a limit to the voltage passing through the bulb. Nevertheless, we concluded with our data that LEDs do not obey Ohm's Law, as they are not resistors but rather diodes, and the difference in data for each LED varied too much for there to be any sort of relationship between variables.
Theory:
How do LEDs Work?
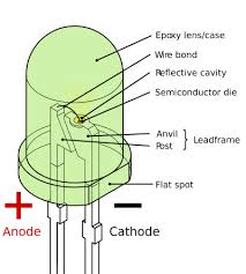
Harkening back to the acronym, the D in LED stands for diode; which is a semiconductor, meaning it can only control current in some cases ("How Semiconductors"). Diode implies that there are two charges in the setup. The diode has a p-side (positive) and an n-side (negative). Both sides are made of small pieces of the metalloid silicon. Silicon is an atom with 4 valence electrons. This means it can stably bond with itself, forming a lattice structure. Since silicon is normally stable, it is not a conductor, but rather an insulator of electric current. Through a process called "doping", the two pieces of silicon to make them have opposite charges. This is done by adding impurities. Small amounts of phosphorus or arsenic, both with five valence electrons, can be added to one part, and the addition of more valence electrons makes the piece of silicon negatively charged. Conversely, elements with three valence electrons like boron or gallium are added to the silicon to introduce a lack of electrons, creating a positive charge. Thus, a diode is created which is able to produce visible light.
LEDs emit light by shooting elections from the n-type side (negative side) across a "p-n junction" to the p-type (positive) side. The p-type side lacks electrons, meaning it is positive and shoots atoms lacking electrons, or holes into the junction. When the two combine, they complete an stable atom and give off a little burst of energy; emitted as a photon. When enough electrons are consumed by holes, they give off enough photons to produce a significant amount of visible light. (Woodford)
The effect of producing light by the process of combining holes with electrons is known as electroluminescence. Electroluminescence is, "...an optical phenomenon and electrical phenomenon in which a material emits light in response to the passage of an electric current or to a strong electric field. This is distinct from black body light emission resulting from heat (incandescence)..." ("Electroluminescence").
The color of light given off by this phenomenon is determined by the energy band gap of the semiconductor in the LED. Electrons can exist in only certain ranges energy states called bands. The valence band is the highest energy range at which a given electron can exist, while the conduction band is the lowest range that an electron can be at ("Band Theory"). A band gap, also known as an energy gap, is, "...an energy range in a solid where no electron states can exist", that is the space between bands ("Band Gap"). If this space overlaps, electrons are able to change energy states easily, meaning that the object is usually a conductor. If the bands are far apart, then electrons cannot easily change energy states and the object is an insulator. On the other hand, if the bands do not overlap but have a small band gap, electrons may still have the chance of changing energy states. This is what creates semiconductors. If the semiconductor is doped, then the electrons have a much easier capability to shift energy levels. The gap can be crossed by electrons that have a potential difference of approximately one volt. However, this can change depending on the material the semiconductor is made with ("Band Theory"). In the case of LED's the electrons at different energy levels will produce different frequencies of light, causing them to produce a variety of colors on the visible spectrum. Below is a table of common semiconductors and the colors they create.
LEDs emit light by shooting elections from the n-type side (negative side) across a "p-n junction" to the p-type (positive) side. The p-type side lacks electrons, meaning it is positive and shoots atoms lacking electrons, or holes into the junction. When the two combine, they complete an stable atom and give off a little burst of energy; emitted as a photon. When enough electrons are consumed by holes, they give off enough photons to produce a significant amount of visible light. (Woodford)
The effect of producing light by the process of combining holes with electrons is known as electroluminescence. Electroluminescence is, "...an optical phenomenon and electrical phenomenon in which a material emits light in response to the passage of an electric current or to a strong electric field. This is distinct from black body light emission resulting from heat (incandescence)..." ("Electroluminescence").
The color of light given off by this phenomenon is determined by the energy band gap of the semiconductor in the LED. Electrons can exist in only certain ranges energy states called bands. The valence band is the highest energy range at which a given electron can exist, while the conduction band is the lowest range that an electron can be at ("Band Theory"). A band gap, also known as an energy gap, is, "...an energy range in a solid where no electron states can exist", that is the space between bands ("Band Gap"). If this space overlaps, electrons are able to change energy states easily, meaning that the object is usually a conductor. If the bands are far apart, then electrons cannot easily change energy states and the object is an insulator. On the other hand, if the bands do not overlap but have a small band gap, electrons may still have the chance of changing energy states. This is what creates semiconductors. If the semiconductor is doped, then the electrons have a much easier capability to shift energy levels. The gap can be crossed by electrons that have a potential difference of approximately one volt. However, this can change depending on the material the semiconductor is made with ("Band Theory"). In the case of LED's the electrons at different energy levels will produce different frequencies of light, causing them to produce a variety of colors on the visible spectrum. Below is a table of common semiconductors and the colors they create.
Why are LEDs More Efficient Than Incandescent Light Bulbs?
Unlike incandescent light bulbs, which emit a broad spectrum of light, including in the infrared spectrum of light which is an indication of heat, LEDs emit light only in the visible spectrum of light. This efficiency comes because almost none of the energy used to power the LED is converted into infrared light. This is because LEDs have a specific band gap, or energy difference between the top of the valence band (semiconductor parts) and the bottom conduction band (insulator parts). Because so much more of the energy is being converted into visible light as opposed to infrared light, LEDs are much more efficient.
The reason that incandescent bulbs are less efficient than LED's is partially due to the nature of the filament used in most incandescent bulbs. Like LED's, incandescent light bulbs work due to an electric current running through them. When the current runs through a coiled tungsten filament in the light bulb, the filament heats up, causing it to glow and produce intense light (Engineer guy). More specifically, the current causes electrons to go into excited state, producing photons which are light particles (Engineer Guy). The electrons are also vibrating, causing heat, which means that only some of the energy produced is visible light. Only about 2.2% of the energy in a standard incandescent light bulb produced by the current becomes visible light (Wikipedia), while the rest becomes infrared lights, which is an indicator of heat ("Incandescent"). The average amount of light produced by an incandescent bulb is about 16 lumens/Watt. What is happening in the light bulb is the resistance is increasing. The flow of current is the flow of electrons freely between atoms. There are some electrons that move and some others that stay in the shells of an atom. Heat causes atoms to vibrate, causing the frequency of collisions between free-moving and captive electrons. This makes it harder for electrons to move through the medium, increasing the resistance of the circuit ("Temperature Effects"). To compare, the band gap that LED's act in is all in the visible light spectrum (Schuster). This means that there is no heat produced by the LED. This gives it lower resistance and the ability to produce visible light much more efficiently, at about 150 lumens per Watt.
The relationship between infrared light produced by the filament and heat is explained by blackbody radiation. The filament has some of the qualities of a blackbody, in that is an object that absorbs most of the radiation around it. However, when it is heated, it emits some light wavelengths out of the visible spectrum, infrared light (Palma).
The reason that incandescent bulbs are less efficient than LED's is partially due to the nature of the filament used in most incandescent bulbs. Like LED's, incandescent light bulbs work due to an electric current running through them. When the current runs through a coiled tungsten filament in the light bulb, the filament heats up, causing it to glow and produce intense light (Engineer guy). More specifically, the current causes electrons to go into excited state, producing photons which are light particles (Engineer Guy). The electrons are also vibrating, causing heat, which means that only some of the energy produced is visible light. Only about 2.2% of the energy in a standard incandescent light bulb produced by the current becomes visible light (Wikipedia), while the rest becomes infrared lights, which is an indicator of heat ("Incandescent"). The average amount of light produced by an incandescent bulb is about 16 lumens/Watt. What is happening in the light bulb is the resistance is increasing. The flow of current is the flow of electrons freely between atoms. There are some electrons that move and some others that stay in the shells of an atom. Heat causes atoms to vibrate, causing the frequency of collisions between free-moving and captive electrons. This makes it harder for electrons to move through the medium, increasing the resistance of the circuit ("Temperature Effects"). To compare, the band gap that LED's act in is all in the visible light spectrum (Schuster). This means that there is no heat produced by the LED. This gives it lower resistance and the ability to produce visible light much more efficiently, at about 150 lumens per Watt.
The relationship between infrared light produced by the filament and heat is explained by blackbody radiation. The filament has some of the qualities of a blackbody, in that is an object that absorbs most of the radiation around it. However, when it is heated, it emits some light wavelengths out of the visible spectrum, infrared light (Palma).
What is Ohms Law?
Some basic resistors, objects that inhibit current flow, follow a formula called Ohm's Law. Ohm law identifies key attributes of a resistor including:
1. Current (I): The amount of charge that goes through the resistor
2. Voltage (V): The change in electric potential energy across a resistor
3. Resistance (R): The constant ratio of voltage to current in a resistive circuit element
The formula for Ohm's Law is:
V = IR
1. Current (I): The amount of charge that goes through the resistor
2. Voltage (V): The change in electric potential energy across a resistor
3. Resistance (R): The constant ratio of voltage to current in a resistive circuit element
The formula for Ohm's Law is:
V = IR
That is, voltage is equal to current times the resistance. It is important to note that this equation can be modeled by a linear graph; it follows the basic linear function equation y = mx+b. In this case, the coefficient would be R, the voltage, and the values of y and x would be the Current and Resistance, respectively. If one were to graph a resistor's voltage compared to its current, one would produce a linear graph with the slope of the line being the resistance.
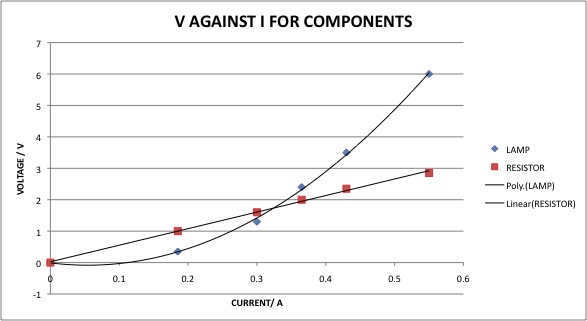
Incandescent light bulbs, on the other hand, do not follow Ohm's law. Due to the amount of excess heat generated by incandescent light bulbs, the resistance of the light bulb increases at an increasing rate as the temperature of the bulb increases. A graph of voltage and current for an incandescent light bulb and resistor looks like this:
Incandescent light bulbs, on the other hand, do not follow Ohm's law. Due to the amount of excess heat generated by incandescent light bulbs, the resistance of the light bulb increases at an increasing rate as the temperature of the bulb increases. A graph of voltage and current for an incandescent light bulb and resistor looks like this:
Since LED's do not generate any excess heat, our hypothesis is that it may have a better chance of obeying Ohm's law, as it would have no heat to alter its resistance. Given the information here, it would be reasonable to assume that it in fact does follow Ohm's law.
For more information on how resistors and traditional incandescent lightbulbs are affected by Ohms Law, refer to the lab report on the matter below:
For more information on how resistors and traditional incandescent lightbulbs are affected by Ohms Law, refer to the lab report on the matter below:
Are LEDs Dimmable?
Traditionally, one common drawback of switching to LEDs is that they, for the most part, cannot be dimmed like traditional incandescent light bulbs can. This is due to how incandescent light bulbs are dimmed, simply by cutting down the amount of voltage going across the bulb smoothly until the bulb completely turns off once the voltage hits 0v.
For most LEDs (such as standalone LEDs or cheaper LED consumer light bulbs), this simply isn't the case. LEDs rely on a constant voltage and a very narrow current margin to stay on without burning out or running into other problems. Because LEDs must keep voltage and current almost constant in order to function, traditional dimmer switches, such as those in most homes, will not work for LEDs.
Over the last few years, engineers have found new ways of dimming LED lights, the most common being with a new technique known as Pulse Width Modulation. According to Elemental LED, "PWM causes the LED to flicker on and off at a rate undetectable to the human eye; the flickers are then staggered depending on the desired amount of light" ("Are LEDs"). This method basically fakes the appearance of the LED dimming by fluctuating between the on and off position of the LED. This technique has become more and more sophisticated over the years, but it can still have significant problems in certain circumstances.
In the TV production or theater world, the flickering of the LED light can become much more visible, especially when the lit area is filmed with a camera that shoots at a low frame rate, such as 24fps. If the LED flickers enough, it can be picked up by the camera as a flickering scene; becoming very disconcerting for the viewer.This may not be a problem in a common household where cameras are less prevalent, but even so, it is not always the best compromise.
Furthermore, most LEDs offer some type of voltage reduction to actually dim the LED instead of using PWM or in tandem with PWM. This method however, will result in the LED abruptly shutting off at a certain voltage, once the LED no longer has enough voltage to continue giving off photons.
Some LED manufacturers, such as Phillips, have tried to make up for these shortcomings by creating LED light bulbs that dim using PWM, but as they dim, the color temperature also starts to change, much like a traditional light bulb would in order to be more natural-looking to the eyes. (Dr. Daniel Smith)
For most LEDs (such as standalone LEDs or cheaper LED consumer light bulbs), this simply isn't the case. LEDs rely on a constant voltage and a very narrow current margin to stay on without burning out or running into other problems. Because LEDs must keep voltage and current almost constant in order to function, traditional dimmer switches, such as those in most homes, will not work for LEDs.
Over the last few years, engineers have found new ways of dimming LED lights, the most common being with a new technique known as Pulse Width Modulation. According to Elemental LED, "PWM causes the LED to flicker on and off at a rate undetectable to the human eye; the flickers are then staggered depending on the desired amount of light" ("Are LEDs"). This method basically fakes the appearance of the LED dimming by fluctuating between the on and off position of the LED. This technique has become more and more sophisticated over the years, but it can still have significant problems in certain circumstances.
In the TV production or theater world, the flickering of the LED light can become much more visible, especially when the lit area is filmed with a camera that shoots at a low frame rate, such as 24fps. If the LED flickers enough, it can be picked up by the camera as a flickering scene; becoming very disconcerting for the viewer.This may not be a problem in a common household where cameras are less prevalent, but even so, it is not always the best compromise.
Furthermore, most LEDs offer some type of voltage reduction to actually dim the LED instead of using PWM or in tandem with PWM. This method however, will result in the LED abruptly shutting off at a certain voltage, once the LED no longer has enough voltage to continue giving off photons.
Some LED manufacturers, such as Phillips, have tried to make up for these shortcomings by creating LED light bulbs that dim using PWM, but as they dim, the color temperature also starts to change, much like a traditional light bulb would in order to be more natural-looking to the eyes. (Dr. Daniel Smith)